The Troodos and Harz mountain ranges have been selected as the geographical framework of the research for the two main thrusts of this archaeometric research.
For the late medieval and early modern period, the Harz mining region, very well documented by authors such as Theophilus Presbyter, Albertus Magnus, Lazarus Ercker, and Erasmus Ebner, and by a huge amount of technical and historical written sources, to such an extent detailed that it provides detailed descriptions, and depictions, of metallurgical practices leading to the formation of zinc-rich compounds as by-products of smelting ad its use in brass cementation since at least the 16th century
These sources point to situations that are closely comparable to those implied by the ancient evidence from Cyprus, both from a geological perspective, later confirmed by historical and modern mineralogical studies in the two regions, and from a metallurgical perspective, in which complex, mixed, sulphide ores represent a primary source for the generation of zinc-rich materials.
Spatial and chronological distance, multiple proximities:
Mining and metallurgical vocation with deep historical roots: Mastering smelting of complex mineral assemblages
Both the Harz and the Troodos mountain ranges developed a strong mining vocation centred on the exploitation of complex sulphide ore assemblages, rich in copper, lead, and zinc. In these contexts, smelting practices were necessarily adapted to ores composed of multiple metallic components, requiring a high degree of technical control over roasting, reduction, and furnace atmospheres. Such conditions favoured the systematic formation of secondary products, including zinc-rich compounds, which became recognisable materials within metallurgical practice, and with experience, also usable in manifold ways.
In Cyprus, the metal-bearing deposits of the Troodos Mountains derive from the formation of volcanogenic massive sulphides associated with an ophiolitic complex, generated by high-temperature hydrothermal circulation at ancient seafloor vents. Fragments of fossil black smokers have been identified within the brecciated massive sulphide bodies of the Troodos complex, producing deposits rich in iron, copper, lead, and zinc, including chalcopyrite and zinc sulphides. These mineralisations occur within the upper portion of the obducted oceanic crust, interlayered with pillow lavas, and are documented at sites such as Skouriotissa, Mathiatis, Agrokipia, and Peristerka. Hydrothermal circulation of acidic, high-temperature seawater favoured metal transport and precipitation in lenses, while later tectonic movements locally overturned the original stratigraphy, bringing deeper mineralised horizons to the surface. Natural gossans, together with mineral exploitation that activated additional acid mine drainage, led to the formation of extensive gossan zones enriched in metals in oxidised and sulphate forms. These mineral assemblages were already recognised in antiquity, as described by Strabo and Galen, and were also probably deliberately exploited. The interaction between natural alteration processes and mining activities contributed to the creation of complex metallurgical landscapes, and these geological transformations induced the development of practices in metallurgy.
In the Harz, part of the mineralisation, mostly at the Rammelsberg, formed through synsedimentary exhalative processes, producing massive sulphide ores characterised by a very fine-grained and intimate admixture of different minerals. These so-called Melierterz, mixed or complex ores, typically include chalcopyrite, galena, sphalerite, and pyrite, intergrown at a scale that prevented effective mechanical sorting. As a result, unwanted components, especially zinc-bearing sphalerite, could not be selectively removed before smelting and inevitably entered the furnace charge. Subsequent tectonic deformation, folding, and faulting further disrupted the original stratigraphy, juxtaposing different ore types within a limited area and contributing to the exceptional geological complexity later described as the “Classic Geological Square Mile”, where a remarkable diversity of mineral assemblages occurs in a very small spatial extent.
These complex mineral resources shaped long, complex and adaptive histories of miners and metallurgists, whose practices and knowledge evolved in close interaction with complex and changing mineral landscapes.
Further related similarities: the recognition of zinc oxide by-products
Kadmeia and Ofengalmei
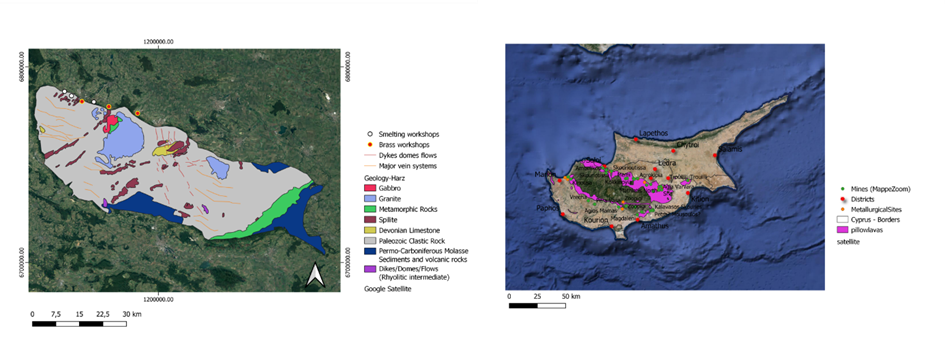
In the ancient period, Pliny the Elder reports the use of cadmea in alloying copper for the production of orichalcum, a process discussed in more detail in the following section. He then returns to cadmea more explicitly in Naturalis Historia (Book XXXIV, chapter 22, §100), describing it as a zinc-bearing material that could occur naturally but also be produced artificially in the furnace, particularly during silver smelting. Pliny notes that this furnace-derived cadmea was lighter and of inferior quality compared to the copper-bearing type and explains that, when heated, zinc oxide sublimates and condenses on the walls of the furnace. He further distinguishes several types of cadmea according to their position of formation and physical appearance: capnitis, the lightest form, attached to the smokestacks and resembling white ash; botrytis, an intermediate, heavier type forming in the central part of the chimney, red or ash-coloured; and plakitis, the heaviest and most impure form, pumice-like in appearance, accumulating at the base of the furnace. Pliny also differentiates subtypes of plakitis, namely onychitis (bluish) and ostracitis (black).
These distinctions reflect a detailed ancient understanding of different zinc oxide products forming within the furnace system and are directly relevant to the period contemporary with the Cypriot evidence.
For medieval and early modern Germany, written sources allow the tracing of zinc-rich metallurgical by-products back to at least the 11th–12th centuries, when Goslar entered the Hanseatic trade network. The earliest records mention materials such as cineres and carbones, likely referring to ash-like and carbonaceous residues enriched in metallic compounds produced during smelting. These early terms reflect the recognition of furnace by-products before a more systematic technical vocabulary was established.
In the mid-13th century, Albertus Magnus, who visited Goslar around 1240, referred to brass production using tutia, most likely imported natural calamine (zinc carbonate). In his Mineral Book (1248), he demonstrates detailed familiarity with the Rammelsberg mine and notes the effect of calamine in turning copper yellowish, while also discussing the role of arsenic in altering copper colour. According to later interpretations, his mention of tutia without naming Goslar as a source indirectly suggests the use of imported zinc minerals rather than local production.
During the 16th century, Georgius Agricola systematised earlier knowledge by recording numerous terms related to tutia and cadmea fornacum. Although he likely did not visit the Harz directly, his works consolidate previous descriptions and terminology circulating within Central European mining culture.
More detailed technical observations come from Lazarus Ercker, who provides explicit descriptions of furnace operation, smelting of complex ores, and the formation of zinc-rich by-products such as Ofengalmei. Ercker worked extensively in the Harz and he also refers to earlier mining knowledge and practices, including those documented by other authors active in the Harz.
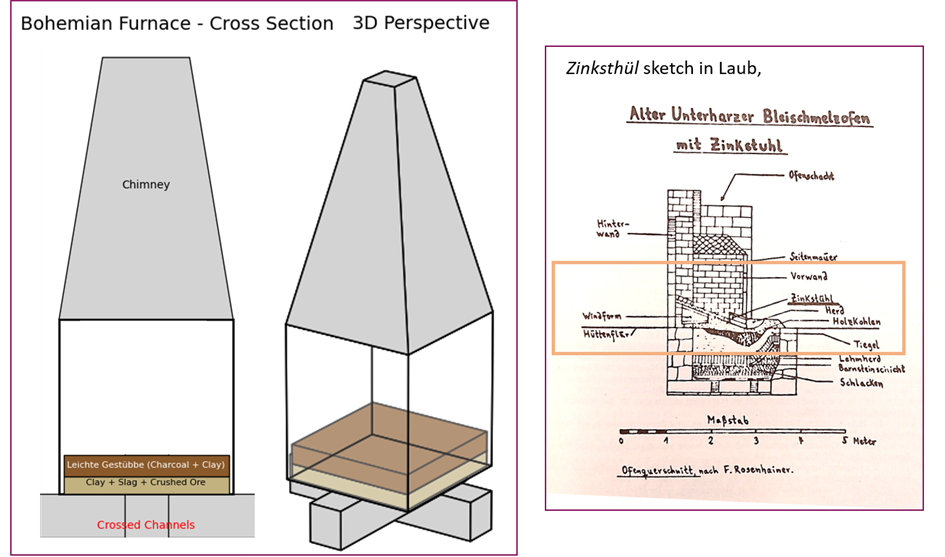
Finally, Erasmus Ebner, associated with the ‘invention’ or better rediscovery of brass cemetation in the Harz with Ofengalmei (16th century), also compiled a comprehensive catalogue of minerals and compounds occurring in or produced by mining activities in the Harz. On cadmia, galmei, or lapis calaminaris, Ebner observed that large quantities of zinc-rich material were lost with the Hüttenrauch (furnace smoke), while heavier material settled near the Rauchloch (chimney opening). His discussions with smelters about constructing vaults above chimneys to improve the recovery of Ofengalmei show a clear concern with maximising by-product yield and reflect an advanced awareness of zinc oxide formation during smelting, and its use for brass production.
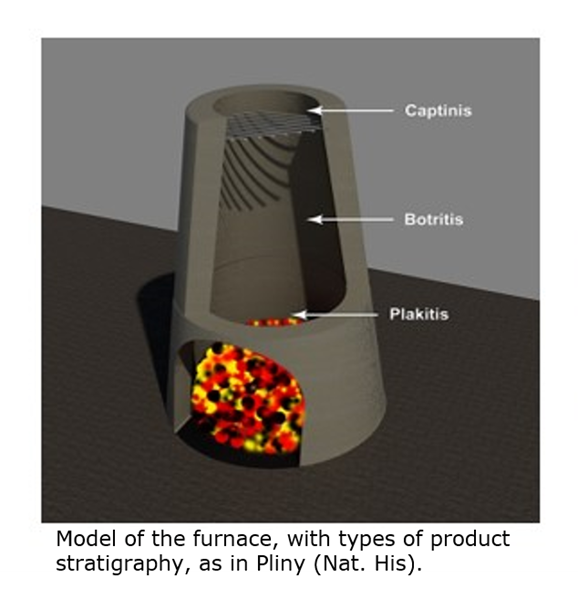
In conclusion, the furnaces of these two places correspond quite well. Dioscorides describes two-storey furnaces in Cyprus designed to promote the sublimation and collection of kadmeia in an upper chamber above the smelting area. Centuries later, Lazarus Ercker illustrates comparable vertically organised furnaces used for the recovery of Ofengalmei in Goslar (Harz), pointing to a shared technological tradition despite the chronological distance.

The rediscovery of this method in the late medieval Harz led to increasingly advanced technological solutions aimed at maximising the recovery of zinc, both as oxide and as metal, culminating in specialised installations such as the Zinkstuhl typical of the Harz area in early modern times. The Bohemian furnace was instead an innovation brought by the experienced Czech metallurgists.
Furnace walls, crucibles and slags from the Rammelsberg
Analytical characterisation to understand zinc behaviours
The study of archaeological materials from storerooms of the local heritage authorities, Niedersächsiches Landesamt für Denkmalpflege (Annexes of Goslar and Braunschweig), including the Klappauf Collection, assembled by Herr Dr. Lothar Klappauf, former director of the NLD Goslar and pioneer of mining archaeology in the Harz. The materials are selected specifically to document the metallurgy of Rammelsberg ores over time. The selected assemblage includes slag fragments, technical refractory ceramics such as crucibles and furnace linings, and lithic materials, such as furnace walls, representing different stages of metallurgical activity, supporting the understanding of thermal and atmospheric conditions.
The investigation aims to understand how zinc behaves and interacts with different substrates over time, helping to identify process fingerprints.
The analysis of the materials followed a strategy of progressive analytical deepening. Initial screening was carried out using pXRF to select the most promising samples, primarily those with higher zinc contents. The selected materials were then examined by optical microscopy and subsequently analysed by SEM–EDS after mounting polished sections in epoxy resin. The results show that, moving from furnace walls, ceramic lining, to crucibles and slags, zinc becomes increasingly intermixed and reactive. The furnace walls and linings are also indeed very sensitive to Zn dynamics. In all these contexts, materials act not only as passive substrates or media, but also as active participants in high-temperature reactions involving zinc.
The comparison between furnace walls and crucibles highlights the complementary nature of these archives. Furnace linings primarily record vapour-phase transport, condensation and diffusion of zinc, effectively acting as a long-term recording of furnace atmosphere. The mineralogical form of zinc can provide indications of furnace atmosphere and function. On furnace walls and linings, zinc occurs mainly as zinc oxide and Zn–Fe oxides, including mixed spinels in which iron is trivalent (Fe³⁺), such as franklinite and zinc ferrite, pointing to oxidising conditions and vapour condensation. Seldom, the presence of copper absorption and lead is more limited to the surface, underlining different properties. In vitrified zones of furnace walls or linings, willemite and fayalite are also documented, indicating points of contact with the high temperature batch. Fragments of linings testify to high temperatures and close contacts to the melt mass, as testified by the presence of copper and lead matte prills and copper droplets. Zinc oxide can be found in superficial, acicular coating, in the outermost layer, typical of crystallisation from vapour, and in layered compact structures below the surface.
Crucibles, by contrast, record different operational roles in melting and cementation, all showing higher zinc absorption in the ceramic fabric, higher melting features, like fractures and vitrification of the ceramic body, where zinc is incorporated into silicates, and thick fuel-ash layers; the crucibles linked to melting or purification display more oxidising signs, including tenorite produced by hot oxidation. More frequent presence of metal prills is also noticed.
In slags, the dominance of mixed silicates such as fayalite–willemite solid solutions can indicate high-temperature melting in reducing environments, where divalent iron (Fe²⁺) is partially substituted by zinc, while the presence of mixed iron-zinc oxides ((Fe, Zn)O) and pure wüstite FeO can indicate stable reducing conditions, in contrast to free iron oxides such as hematite, which mark locally oxidising zones. The presence of matte system phases is predominantly associated with slag but also documented in crucibles, reflecting incomplete desulphurisation of complex sulphide charges in smelting, the use of black copper or cementation with sulfidic-rich compounds such as mixed ores or sulphur-rich Ofengalmei.
The mineralogical form of zinc can provide indications of furnace atmosphere: zinc oxides and Zn–Fe oxides point to oxidising conditions and possible vapour condensation processes, whereas zinc incorporated into silicates or retained within sulfidic phases is often associated with more reducing high-temperature conditions.
These analyses gave the chance to have a closer look at archaeological materials connected to zinc-rich ores (in the case of furnaces and slag fragments) or compounds in the case of cementation crucibles, and zinc-rich alloys in the case of melting or refining crucibles. Additionally, they had the chance to observe the materiality and appearance of these samples closely. In the case of the slags, the high zinc contents resulted in the presence of cold white layers inside the slag body and on the outside of them, to which also contributed postdepositional actions. Same whitish aspect, or grey/lilac can be observed on furnace walls, as also experimental trials helped to evidence. These observations have been used to carry out survey missions addressed to the collection of zinc-related materials.

Examples of zinc absorption in furnace walls (445, 448) and in crucibles related to cementation procedures carried out in medieval worshops of Braunschweig.



